Drug-resistant Tuberculosis accounts for 1 in 3 deaths from antimicrobial resistance (AMR)*, meaning grim predictions for a ‘post-antibiotic age’ are already a reality for many countries. This is why a ‘test-of-cure’ diagnostic to extend the life of new drugs is so urgently needed, AMR expert Dr Ben Swift, R&D Director for PBD Biotech, commented at BioInfect 2024.
Dr Swift says: “Our phage-based diagnostic has potential to monitor the efficacy of drug treatment – something not possible with current technology – as well as offering the opportunity to identify antibiotic-resistant TB.”
He explains that AMR occurs when microorganisms causing disease no longer respond to the drugs. “One of the reasons that AMR has increased for TB is that the traditional treatments require multiple drugs, taken over extended time periods. It is therefore difficult for many communities to gain access to the right medication, and patient compliance can be low as the drugs have severe side-effects.”
Tuberculosis disease is fatal if untreated and only about 2 in 5 people with drug resistant TB accessed treatment in 2022. A further 3.1 million cases remain undetected each year. The most widely used test for TB relies on measuring the patient’s immune response, which occurs when the disease is well progressed; the test also cannot distinguish between active disease and past infection.
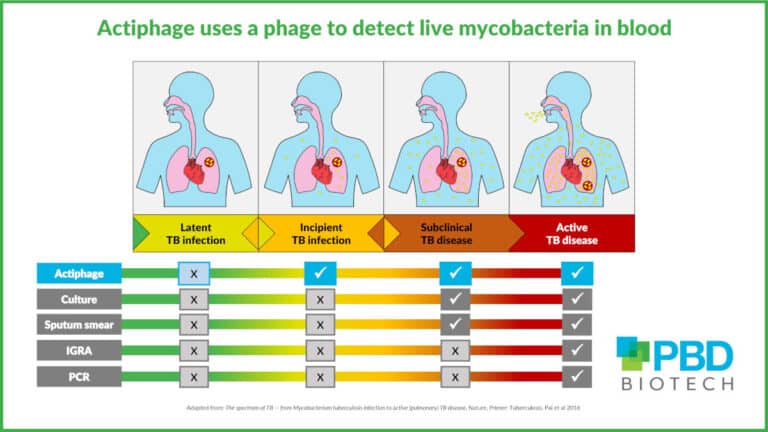
PBD Biotech has developed Actiphage TB, which takes advantage of the specificity of interaction between a phage and its host bacteria to create a rapid diagnostic for TB and other diseases caused by Mycobacteria.
Dr Swift continues: “Our powerful technology uses a phage – the natural enemy of bacteria – to hunt down live bacterium and lyse them, to allow the DNA to be identified with qPCR.
“We can detect Mycobacterium tuberculosis (Mtb) in a simple blood sample within a day. The current culture method takes up to eight weeks to grow Mtb from a sputum sample.”
He explains that Actiphage, which is currently in clinical trials in Leicester, UK, would offer a new tool to fight AMR through improved diagnosis and test-of-cure.
Actiphage is also in lab user trials by the R2D2 Network in the Philippines. The country has a high incidence of TB and, in 2021, was one of the first to adopt BPaL, a new short-protocol medicine for multidrug-resistant TB.
Dr Swift continues: “BPaL is a six-month treatment that has been hailed as a breakthrough in the fight to End TB. In parallel with the development of new drugs, it is vital that new diagnostics are developed to target them effectively.
“By making the bacterial DNA available for sequencing, Actiphage would enable an informed choice of treatment for the patient, reducing the risk of resistance. Additionally, as Actiphage detects only live mycobacteria it can also be used after the course of treatment as a test-of-cure and to support the development of new drugs.”
Dr Swift discussed the role of phage-based TB diagnostics in the ‘What’s in the pipeline’ session of BioInfect 2024 on Thursday 8th February 2024 at The Spine, Liverpool L7 3FA.
* https://www.tballiance.org/why-new-tb-drugs/antimicrobial-resistance