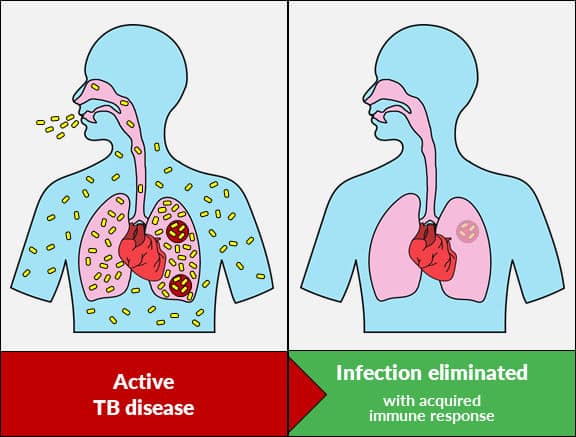
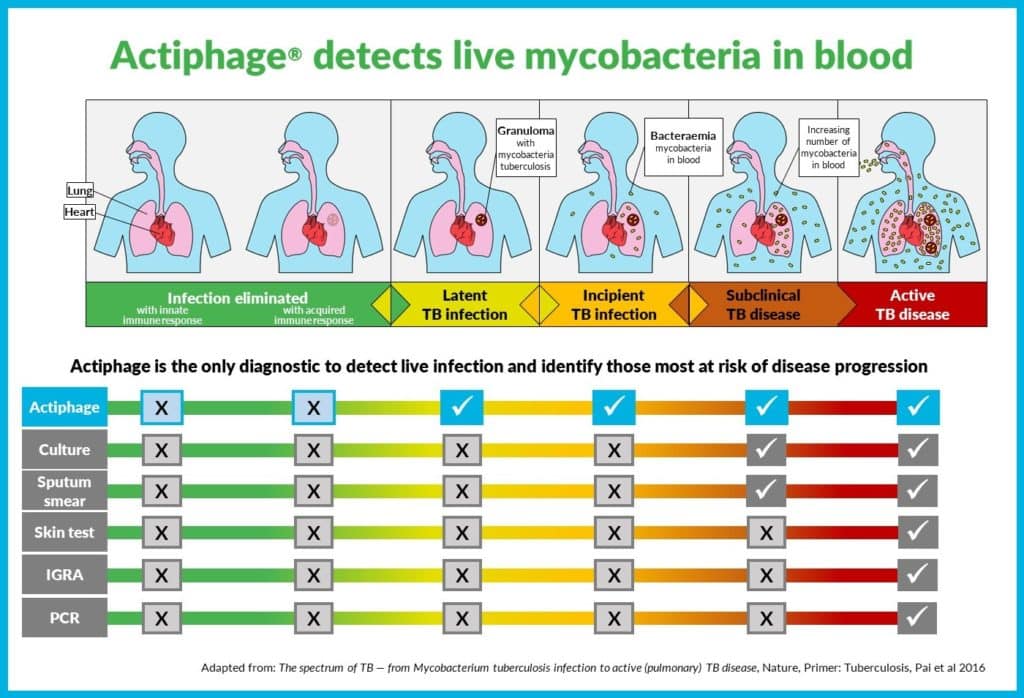
Through detection of active Mycobacteria across the spectrum of tuberculosis infection Actiphage offers unique role in TB testing
Role in TB testing
- There are currently no reliable biomarkers for the detection of incipient TB, despite research to identify new TB biomarkers and diagnostics.
- Immune-based diagnostics provide evidence of an immune memory response to TB infection rather than confirming the presence of active organisms.
- Traditional PCR-based methods do not discriminate between active, viable and non-viable mycobacterium tuberculosis.
- Actiphage is the only blood-based test that combines phage technology to detect only active, disease-causing mycobacteria with traditional qPCR methods.
- Actiphage does not require cultivation to detect bacteria and has higher accuracy and faster turnaround times.
Tuberculosis Drug Treatment Management
Preventive Drug Therapy
- Current immune-based LTBI diagnostics (TST, IGRA) have a poor positive predictive value (PPV), so targeted testing and treatment is only recommended for high risk groups.
- Actiphage is designed to detect incipient TB and disease progression, which may allow for scale-up of preventive treatment beyond the current risk groups.
- Actiphage is a blood-based test designed for screening of LTBI in populations with an easily collected sample.
- Actiphage may create a significant opportunity for LTBI preventive therapy drug development.
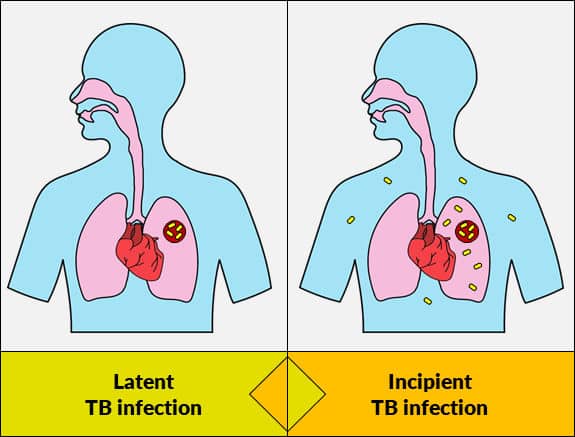
TB Drug Monitoring
- Actiphage has the potential to be a diagnostics marker for drug response and predictors of relapse.
- Actiphage provides confirmation of active Mtb and may overcome certain barriers to LTBI therapy such as uncertainty of diagnosis.