Actiphage is the first test to confirm TB disease progression by detecting the live pathogen in the blood. By supporting targeting of treatment, it offers the opportunity to #EndTB. For this, its developers PBD Biotech have been named as one of six semi-finalists in the ADLM 2024 Disruptive Technology Award.
Lack of effective TB diagnostics
A quarter of the world’s population are carriers of tuberculosis, of which 10.6 million will get ill each year due to a lack of effective diagnostics.
Recent research by Amsterdam UMC, showed that 80 percent of people with active tuberculosis have no cough – the entry point for diagnosis. This leaves a huge reservoir of disease in the population. The lack of effective diagnostics to detect TB in its early stages is one of the reasons why this preventable, curable condition is the world’s most lethal infectious disease.
Test of cure needed to overcome AMR threat
Drug-resistant TB also accounts for 1 in 3 deaths from antimicrobial resistance, which is why a test of cure to extend the life of new drugs is so urgently needed.
Actiphage addresses both these unmet needs and is being developed to meet the World Health Organisation Target Product Profile (TPP) for tuberculosis. Ending the TB epidemic is Sustainable Development Goal Target 3.3.
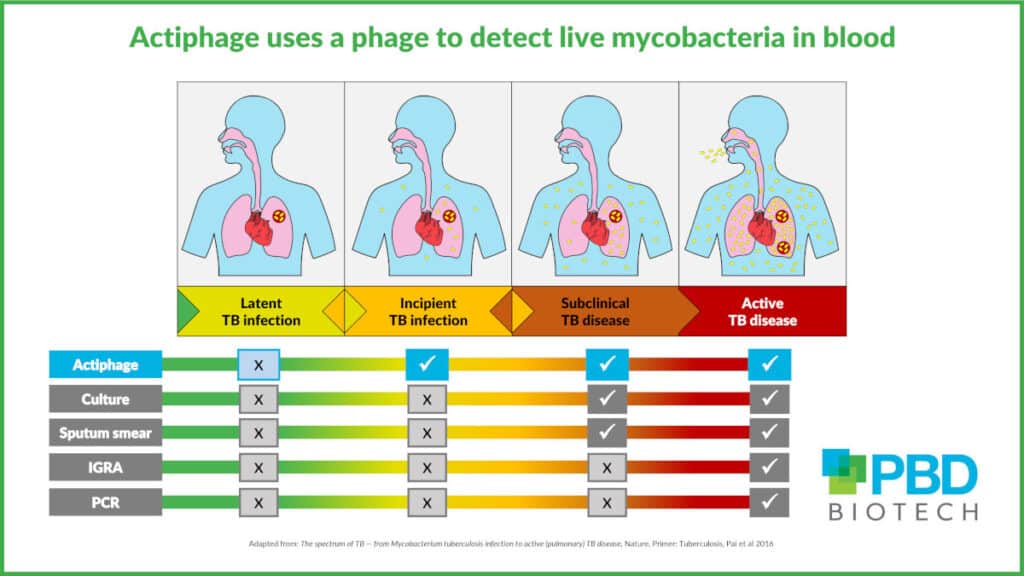
Bacteria in blood early indicator of active TB
Recent clinical trials have confirmed that bacteraemia is a reliable indication of active TB disease progression.
Actiphage is the only diagnostic listed in the TAG Pipeline Report that uses a phage to directly detect live bacteria in the blood. It therefore has potential to be used to screen vulnerable populations to determine which household contacts are at risk of developing the disease and to provide a test of cure following treatment.
Neither of these actions are possible with current tests. Other tests used to screen for TB – such as IGRA – measure the host’s immune response to past exposure to infection, not the current status of the disease.
Jane Theaker, CEO of PBD Biotech, comments: “We are delighted to be shortlisted for this prestigious industry award. Ending the TB epidemic is an international goal, and no country is immune from the impact of this disease. Actiphage offers potential to break the cycle of infection and ADLM’s support in gaining wider profile with the test and treat community is greatly appreciated.”
Patents in UK, US and Africa
Actiphage has gained patents in the US, UK, West Africa, and South Africa. The results of a ground-breaking clinical trial have been announced in Lancet Microbe (2024) and showcased in the Hot Topics session at the 2022 32nd ECCMID congress.
It is currently being reviewed by the R2D2 Network in the Philippines and in clinical trials in the UK and Zambia.
ADLM 2024 Disruptive Technology Award
The ADLM 2024 Disruptive Technology Award Competition (DTAC) aims to evaluate and elevate companies developing new diagnostic tools and technologies that have the potential to improve patient care. DTAC grants the diagnostic and laboratory medicine community an early look at innovative and impactful products that address current gaps in the field and/or improve upon existing solutions. The finals are to be held at the ADLM conference.





