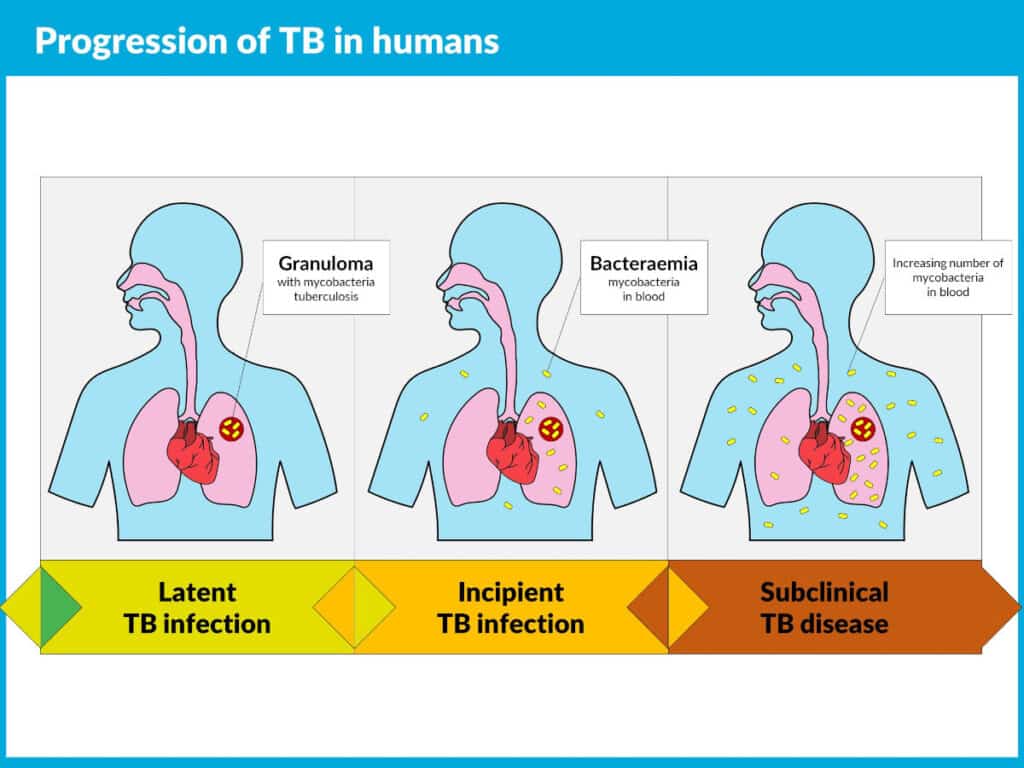
On average, 5–10% of people who are infected with TB will develop disease over the course of their lives, creating a driver for TB drug research.
Early diagnosis of TB, systematic screening, preventive treatment of persons, and vaccination against TB are some of the key components in the WHO pillars for the End TB strategy.
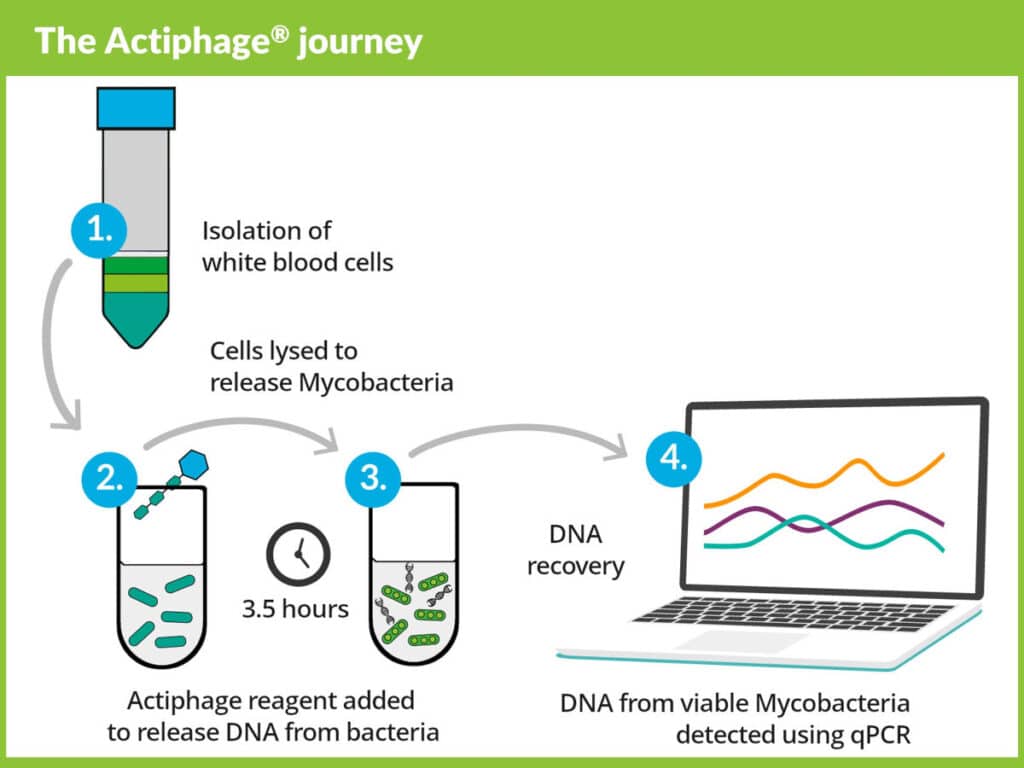
Actiphage plays a unique role in the detection of mycobacterium tuberculosis (Mtb) to potentially allow rule-in of infected individuals for preventive therapy and rule-out for TB vaccination.
Support for TB drug research
- Ground-breaking science – clinical studies have shown promise in predicting which individuals with latent tuberculosis might progress to active disease.
- Companion diagnostic – Actiphage may simplify drug discovery, accelerate clinical study timelines, reduce patient sample size, lower overall costs, and expedite regulatory approval.
- Targeted Testing & Treatment – Actiphage distinguishes viable, active mycobacteria, potentially supporting preventive drug treatment or whether the patient is a candidate for vaccination.
- Personalized Medicine – Actiphage may facilitate prescribing according to strain, health status, and genomics.
Contact Us
General contact form