When combined with qPCR methods, phage-based technologies offer a highly specific and sensitive approach to developing pathogen-directed biomarker for TB (tuberculosis), concludes Dr Pranabashis Haldar in his article for the Pathologist, “Bacteriophage Boost for TB Management”, in which he discusses findings from recent research using the phage-based diagnostics Actiphage TB.
He continues: “The existing diagnostics for TB are inadequate to meet the minimum WHO target product profile thresholds to support disease elimination – proving that faster, more sensitive tests are required, that do not rely on sampling sputum from the site of infection.”
Meeting the WHO target product profile
Dr Haldar explains that host immune biomarkers can inform us of the presence of current or previous TB infection, but cannot discriminate between the two – nor do they tell us anything about the infecting pathogen.
“Only recently have we been able to demonstrate that Mtb is detectable in the blood of people with active or latent TB in the absence of immunodeficiency*,” he explains, as historically, culture-based methods only detected Mtb in the blood of those with very severe illness. The detection of live mycobacteria circulating in the blood provides an early indication of TB disease progression.
Actiphage TB, a novel blood-based biomarker, is being developed to meet the requirements of the WHO target product profile (TPP).
Blood-based, pathogen directed biomarker for TB
Using Actiphage, a phage-based diagnostic, it will become increasingly possible to detect Mtb in the blood of a large number of people with TB infection – supporting future development of effective blood-based, pathogen-directed biomarkers that are low-cost, rapid, and potentially deployable in low-income settings.
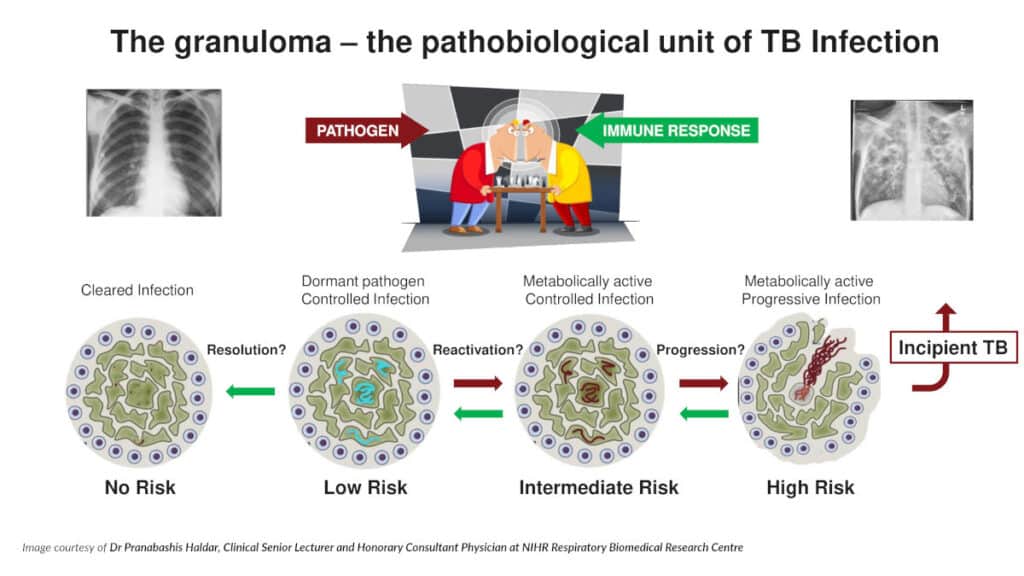


In the Pathologist article, Dr Haldar concludes: “Pathogen-directed biomarkers may complement current host immune markers for evaluating TB infection phenotypes, allowing us to discriminate between individuals who do – and potentially don’t – require treatment for their TB infection. This combination is a powerful tool to have in our arsenal, if we are to drastically reduce TB cases and deaths by 2030.”
Dr Pranabashis Haldar, is a Clinical Senior Lecturer at the University of Leicester, UK and Principal Investigator in the Respiratory and Infection Theme of the Leicester NIHR Biomedical Research Centre, UK.
*PET-CT-guided characterisation of progressive, preclinical tuberculosis infection and its association with low-level circulating Mycobacterium tuberculosis DNA in household contacts in Leicester, UK: a prospective cohort study. Jee Whang Kim el al https://doi.org/10.1016/S2666-5247(23)00289-6


