Emerging interest in the specificity and sensitivity of phage therapy and diagnostics has prompted an increasing need to further understand the physical interactions between phage and host. Whether using phage for therapy or diagnostics, one of the key steps is determining the multiplicity of infection (MOI).
You can view the poster here, and read a summary below.
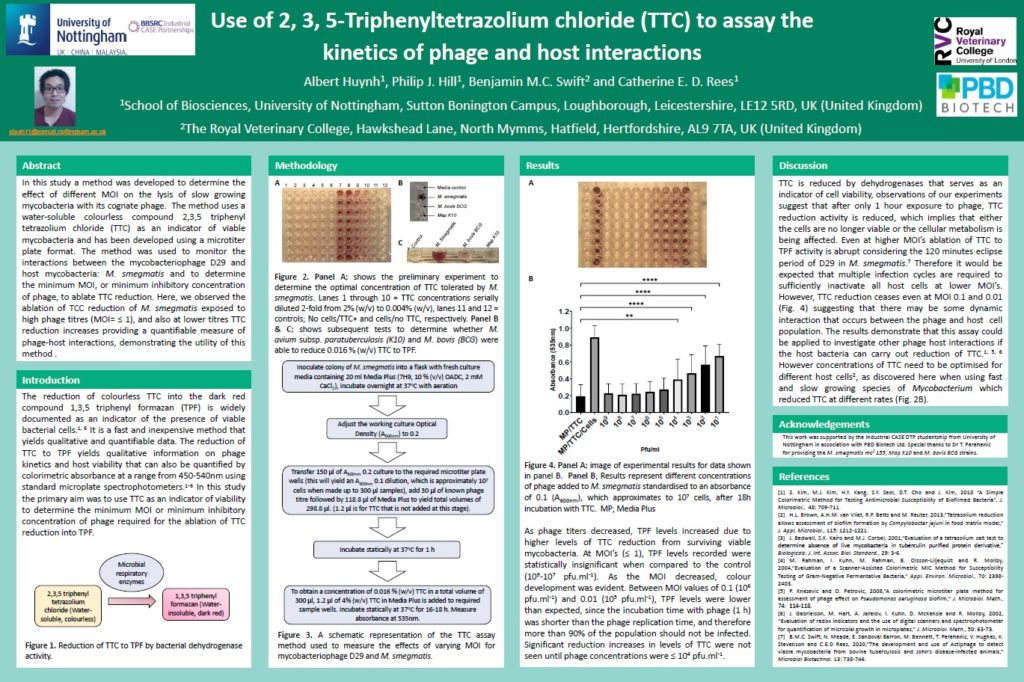
Therefore, in this study a method was developed to determine the effect of different MOI on the lysis of slow growing mycobacteria by its cognate phage, using a microtiter plate format and a water-soluble compound 2, 3, 5- triphenyl tetrazolium chloride (TTC) as an indicator of viable mycobacteria. This method utilises the reduction of TTC into the dark red compound triphenyl formazan (TPF). It is a rapid, inexpensive, and simple method, which yields results that can be both quantified and qualitatively observed. It is noted that the fast and slow growing species of Mycobacterium tested (M. smegmatis, M. avium subsp. paratuberculosis and M. bovis BCG) reduce TTC to TPF at varying rates and have varying tolerances to the concentration levels of TTC and therefore the assay needs to be optimised to accommodate variations in host cell biology.
The colorimetric reduction of TTC to TPF yields qualitative information on phage kinetics and host viability that can also be quantified by colorimetric absorbance at 540 nm that can be monitored using standard microplate spectrophotometers. The method was used to monitor the interactions between the mycobacteriophage D29 and host mycobacteria: M. smegmatis, M. avium subsp. paratuberculosis and M. bovis BCG and was utilised to determine the minimum MOI or minimum inhibitory concentration of phage required to ablate TTC reduction to TPF. Here, we observed the ablation of TCC reduction by M. smegmatis exposed to high phage titres (MOI= ≤ 1), and at lower titres TTC reduction increases, demonstrating the useability of TTC to study phage host interactions.
Read more about the conference and the other papers presented on the Phages 2021 website.