A clinical trial of Actiphage®, a promising new diagnostic for tuberculosis infection, has shown for the first time that live bacteria can be detected in the blood of people with incipient TB infection. Offering almost three times greater specificity than the gold standard IGRA test for this state of latent infection. The ground-breaking clinical trial shows the potential for using Actiphage as a pathogen directed biomarker to identify those with incipient TB, a key WHO research priority.
Nearly a quarter of the world’s population has latent tuberculosis infection (LTBI), which is asymptomatic and a reservoir for future disease. A small proportion have incipient TB, defined as a state of higher risk of developing to TB unless treated. However, separating these individuals from everyone else with LTBI is currently problematic.
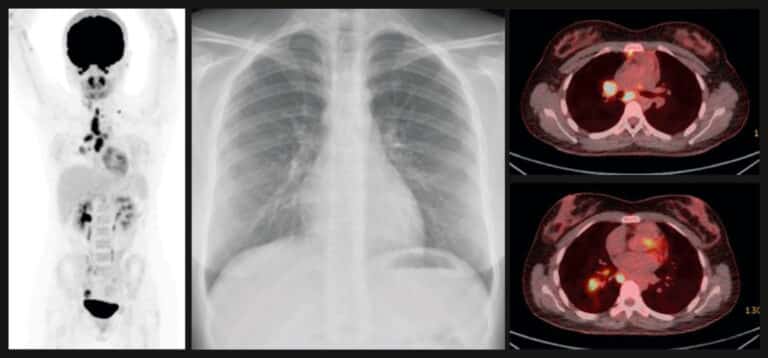
Research, to be presented at ECCMID 2022 (European Congress of Clinical Microbiology & Infectious Diseases) by researchers from the University of Leicester, has shown that Actiphage, a new blood test for Mycobacterium tuberculosis (Mtb) can improve identification of those at risk of progressive tuberculosis infection, offering potential for a rapid, non-invasive biomarker for early screening of asymptomatic individuals.
Lead author, Jee Whang Kim of the Leicester Tuberculosis Research Group (LTBRG), will present the findings of the study in the ‘hot topics in tuberculosis’ session at ECCMID 2022 at 13:30 -14:30 on Tuesday 26th April.
Kim explains: “The trial aimed to investigate whether there is an association between the presence of Mycobacterium tuberculosis (Mtb) circulating in the blood of individuals who are otherwise completely well, and evidence of progressive TB infection.
“The results show that by using Actiphage, we were successful in isolating Mtb in the blood of a subset of individuals with latent TB and also in demonstrating an association with radiological and microbiological evidence of progressive TB infection. Compared with IGRA, Actiphage was almost three times more specific at identifying this group.
“To our knowledge, this is also the first time that viable Mtb infection has been isolated in people with a clinical diagnosis of latent TB infection.”
Dr Pranab Haldar from the University of Leicester is the lead clinician on the trial; he comments: “Although TB is now second to Covid as the leading cause of death from an infectious disease, the future impact of TB on human health is likely to far exceed the Covid pandemic. TB prevention through identification and treatment of at-risk LTBI is a key long-term strategy for TB control and elimination.”
“Our findings demonstrate the potential utility of Actiphage as a pathogen directed biomarker for improving risk stratification of LTBI that can potentially complement the evolving panel of host-directed immune biomarkers.
“We hope our findings will stimulate further research to develop other pathogen directed biomarkers for this purpose.”
Jee Whang Kim presented findings of the study in the ‘Hot topics in tuberculosis’ session at ECCMID 2022 on Tuesday 26th April the extract is available here: “A novel bacteriophage-based assay stratifies tuberculosis risk in recent household contacts of pulmonary tuberculosis: a prospective observational cohort study’
The research team have also published in the Lancet Microbe.